ISO-Aligned Quality Processes
All medical parts manufactured under rigorous quality inspection aligned with ISO 9001 principles and controlled protocols.
Forge Labs partners with medtech innovators to deliver high-performance prototypes, functional components, and short-run production parts using industrial 3D printing—all under ISO-aligned processes and cleanroom handling capabilities. We support biocompatible materials and traceable manufacturing for regulated medical device production.

Healthcare moves fast—and medical device innovation demands rapid prototyping, precision engineering, and materials that meet strict performance and biocompatibility criteria. At Forge Labs, we help bridge the gap between concept and production with advanced additive manufacturing solutions tailored for the healthcare sector. From ventilator components and surgical prototypes to diagnostic housings and wearable enclosures, our experience spans early-stage design validation to functional parts that meet rigorous inspection standards.
Forge Labs helps teams move from design validation to production-ready parts without waiting on tooling lead times. Consolidated assemblies, lighter parts, and faster revisions give engineering teams more control over schedule, cost, and performance.




Medical device work depends on controlled material selection, documented inspection, and traceable handling. We support those requirements across prototyping, verification builds, and pilot manufacturing.
All medical parts manufactured under rigorous quality inspection aligned with ISO 9001 principles and controlled protocols.
USP Class VI certified materials with comprehensive biocompatibility testing and documentation for medical device applications.
Controlled environment processing available for critical medical applications requiring contamination-free manufacturing and handling.
Complete dimensional inspection reports, material certifications, and batch traceability documentation provided upon request.
From concept models to production-intent hardware, each workflow is tuned for the precision, repeatability, and engineering confidence that medical device manufacturing teams demand. Our capabilities span rapid prototyping with sub-millimeter accuracy, complex geometry optimization for weight reduction, and low-volume production with full material traceability — giving engineering teams the speed to iterate faster and the data to qualify parts with confidence.
Stereolithography (SLA) with USP Class VI biocompatible resins accelerates medical device development with functional prototypes suitable for clinical evaluation and human factors testing. Tolerances of ±0.25% (min ±0.25 mm) support precise fit requirements for surgical instruments and patient interfaces. High-definition surface finish eliminates post-processing for visualization studies, while multiple biocompatible material options enable testing across device applications. Digital design iteration reduces development timelines from months to days, supporting rapid innovation cycles critical in medical device development.
SLA Tolerances
±0.25% (min ±0.25 mm)
Biocompatible
USP Class VI resins
Turnaround
24-48 hours
Surface Finish
High-definition
Relevant technologies
Stereolithography (SLA)/Fused Deposition Modeling (FDM)/PolyJet Matrix (PolyJet)/Selective Laser Sintering (SLS)
Material options
High Temperature Resin/Accura 25/Accura Xtreme White 200/Accura ClearVue

Medical Device Prototyping
Accura ClearVue and specialized optical resins provide exceptional transparency and dimensional stability for diagnostic equipment housings, light guides, and optical assemblies. Materials achieve >95% light transmission while maintaining biocompatibility for patient-contact applications. Precision manufacturing ensures critical optical alignment features maintain tolerances throughout sterilization cycles. Custom geometries optimize light path efficiency and minimize optical losses in portable diagnostic devices. Post-processing options include optical polishing and anti-reflective coatings for enhanced performance.
Light Transmission
>95%
Biocompatible
USP Class VI
Tolerances
±0.25mm optical features
Sterilization
Cycle stable
Relevant technologies
Selective Laser Sintering (SLS)/Multi-Jet Fusion (MJF)/Direct Metal Laser Sintering (DMLS)/Fused Deposition Modeling (FDM)
Material options

Diagnostic & Imaging Equipment Components
Selective laser sintering (SLS) with medical-grade PA12 nylon produces complex respiratory components with smooth internal airflow channels and precise dimensional control. Material properties include chemical resistance to disinfectants, steam sterilization compatibility, and excellent fatigue resistance for cyclic pressure applications. Internal channel geometries optimize laminar airflow while minimizing pressure drops across manifolds and valve assemblies. No support structures required enables complex geometries including integrated filters, flow sensors, and pressure relief features impossible with traditional manufacturing.
Channel Tolerance
±0.3mm
Material
Medical PA12 SLS
Sterilization
Steam compatible
Pressure Rating
50+ PSI
Relevant technologies
Selective Laser Sintering (SLS)/Multi-Jet Fusion (MJF)/Direct Metal Laser Sintering (DMLS)/Fused Deposition Modeling (FDM)
Material options

Respiratory & Ventilator Components
Flexible TPU materials and lightweight nylon enable patient-specific wearable device prototypes with anatomical conformance and comfort optimization. Flexible shore hardness options (70A-95A) accommodate varying compression requirements for monitoring devices, therapeutic equipment, and prosthetic interfaces. Design iteration supports anthropometric optimization across patient populations while maintaining consistent performance characteristics. Biocompatible formulations ensure skin compatibility for extended wear applications in research and clinical validation studies.
Shore Hardness
70A-95A TPU
Biocompatible
Skin contact safe
Weight
40-60% lighter than silicone
Flex Cycles
100K+ without failure
Relevant technologies
Stereolithography (SLA)/Fused Deposition Modeling (FDM)/PolyJet Matrix (PolyJet)
Material options
High Temperature Resin/Accura 25/Accura Xtreme White 200/Accura ClearVue
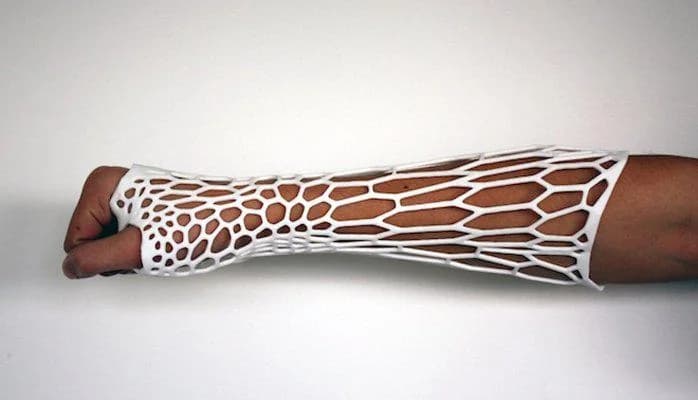
Patient-Specific & Wearable Devices
ISO 9001-aligned quality management systems support cleanroom manufacturing for medical device pilot production and clinical trial components. Environmental controls maintain Class 10,000 (ISO 7) cleanliness standards with documented contamination control procedures. Material handling protocols ensure comprehensive process documentation from raw powder through final packaging with detailed documentation packages. Statistical process control monitoring validates dimensional consistency across production batches while maintaining full chain of custody for regulatory compliance. Cleanroom protocols accommodate volumes from 10 to 10,000+ parts with scalable quality assurance.
Cleanroom Class
ISO 7 (10,000)
Quality System
ISO 9001-aligned
Lot Traceability
Raw to finished
Production Volumes
10-10,000+ parts
Relevant technologies
Selective Laser Sintering (SLS)/Multi-Jet Fusion (MJF)/Direct Metal Laser Sintering (DMLS)/Fused Deposition Modeling (FDM)
Material options

Cleanroom Pre-Production Runs
High-precision surgical guides and clinical instrumentation manufactured with dimensional accuracies suitable for patient-specific surgical planning and training applications. ULTEM and biocompatible materials withstand repeated sterilization cycles while maintaining dimensional stability and surface quality. Custom patient-specific geometries enable precise surgical placement of implants, optimize access angles, and reduce procedure time. Training models provide realistic tactile feedback with appropriate material properties for skill development and procedural validation in medical education programs.
Dimensional Accuracy
±0.25mm
Materials
ULTEM, biocompatible
Sterilization
Multiple cycle stable
Patient-Specific
CT/MRI derived
Relevant technologies
Stereolithography (SLA)/Fused Deposition Modeling (FDM)/PolyJet Matrix (PolyJet)
Material options
High Temperature Resin/Accura 25/Accura Xtreme White 200/Accura ClearVue

Surgical & Clinical Tools

Medical Device Prototyping
Projects focused on airflow-critical components, diagnostic housings, and regulated medical hardware development.

Case Study
Rapid prototyping and production of critical ventilator manifold components during global health crisis. Delivered functional parts with smooth internal airflow channels and biocompatible materials, enabling accelerated device deployment and patient care.
Key outcomes

Case Study
Custom optical housing for portable diagnostic device featuring integrated light guides and precise optical alignment features. Achieved optical clarity requirements while maintaining impact resistance for field use applications.
Key outcomes
Qualified materials selected for performance, certification requirements, and repeatable production in medical device manufacturing environments.

Medical Device Manufacturing grade
Crystal clear biocompatible resin

Medical Device Manufacturing grade
Biocompatible engineering grade

Medical Device Manufacturing grade
Medical grade flexible polymer

Medical Device Manufacturing grade
High-performance thermoplastic

Medical Device Manufacturing grade
Medical grade polycarbonate

Medical Device Manufacturing grade
Medical grade ABS
Compare certifications, mechanical properties, and application fit across our full portfolio before locking your production material stack.
Evaluate each process by geometry freedom, material options, and throughput so you can pick the right path from prototype to production.
Our application engineers can map your requirements to the right technology, material, and finishing workflow before you commit to a build.
Upload your CAD files for an instant quote or speak with our application engineers about your specific requirements.
Upload your CAD file
STL, STEP, OBJ, 3MF supported
Instant pricing and manufacturability feedback