Additive manufacturing has moved from a niche prototyping method to a practical production workflow in medical devices. Teams now use 3D printing to deliver patient-specific geometry, reduce development cycle time, and improve fit confidence in applications where dimensional reliability and clinical performance matter.
The medical device manufacturing sector operates under constraints that are harder than most product categories: strict quality systems, traceability expectations, biocompatibility requirements, and little tolerance for fit or function errors. Within those constraints, additive processes provide one major advantage over legacy methods: they preserve design freedom while still supporting manufacturing discipline.
This article outlines where 3D printing is delivering measurable impact today, how teams use it during regulated development, and which emerging capabilities are most relevant to next-generation device programs.
Scope of Impact
- Patient-specific external devices and interfaces
- Surgical planning models and procedure guides
- Prototype-to-production transitions for low and medium volumes
- Faster design verification before expensive tooling decisions
Patient-Specific Device Development
The most established benefit of additive manufacturing in healthcare is anatomical customization. Conventional tooling-based processes are optimized for repeatable standard geometries, while many medical applications require geometry that conforms to individual anatomy. 3D printing closes that gap by allowing device geometry to be generated directly from scan-informed CAD data.
| Technology | Typical Medical Use | Why Teams Choose It |
|---|---|---|
| SLA | Surgical guides, anatomical models, hearing aid shells | High resolution and smooth feature definition |
| SLS / MJF | Orthotics, prosthetic components, functional housings | Durable nylon parts with no support structures |
| DMLS | Metal implants and surgical tool components | Complex geometry with high-performance alloys |
Prosthetics and Orthotic Interfaces
Prosthetic development has shifted toward lightweight, patient-specific structures that combine functional performance with personalized fit and aesthetics. Additive manufacturing enables this by supporting rapid geometry iteration without new tooling, which is especially important when fit adjustments are required after initial patient trials.

Patient-specific prosthetic geometry can be revised quickly without retooling.
Hearing and Dental Applications
Hearing aids and dental appliances are two of the clearest proof points for additive at scale. Both categories depend on personalized geometry, and both have demonstrated high-throughput digital workflows from scan capture through finished part production. This is where additive is no longer experimental; it is the default operating model.
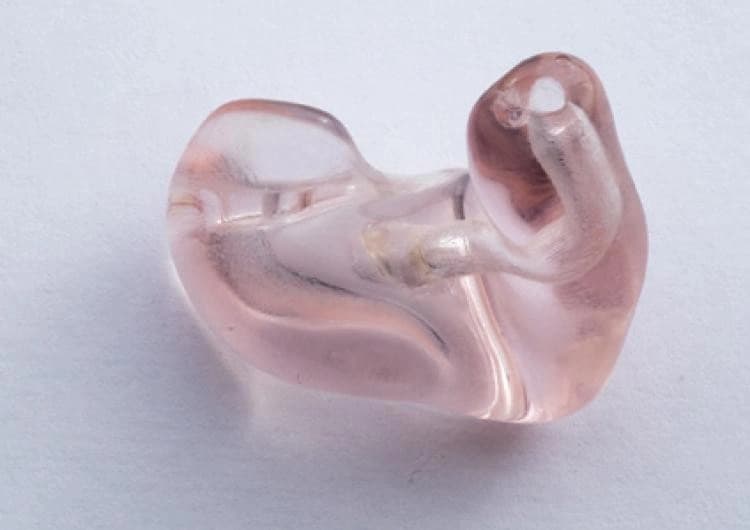
Hearing aid manufacturing is a mature example of additive production at medical scale.
In dental workflows, printed models and appliance components help reduce turnaround time compared with legacy lab methods and improve predictability in fit-driven products.
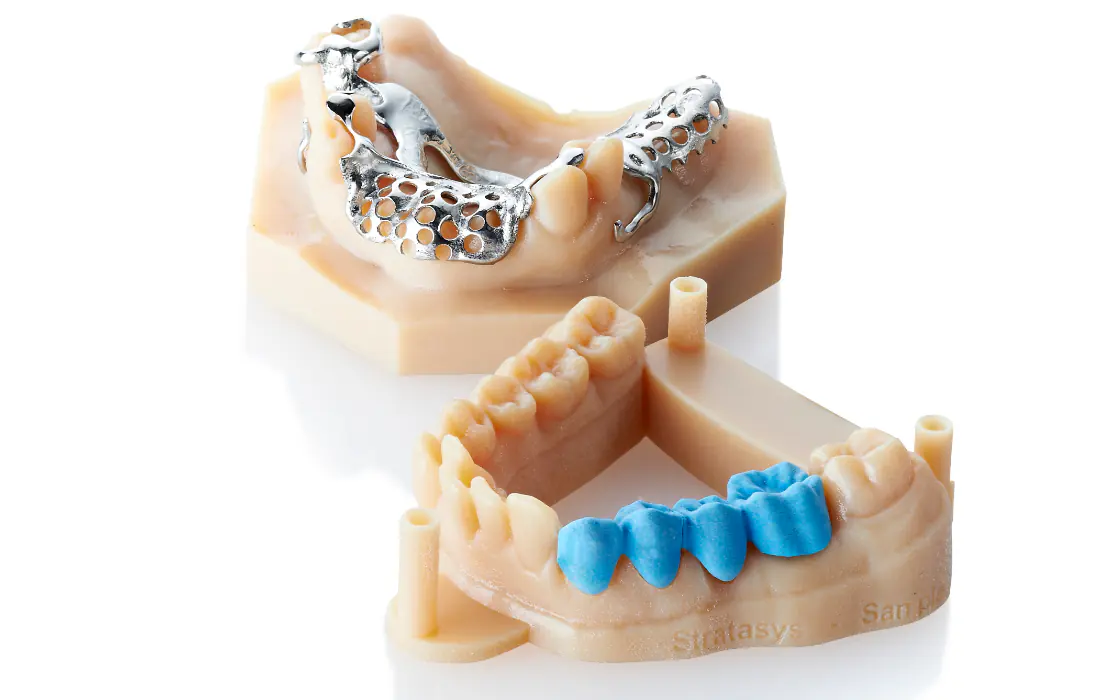
Medical and dental model printing supports treatment planning and fit verification.
Rapid Prototyping in Regulated Programs
Speed in medical product development is not just about shipping faster. It is about reducing uncertainty before design freeze, validating ergonomics and assembly behavior earlier, and preventing late-stage rework that can affect quality documentation and submission timelines. Additive manufacturing helps by compressing the design-build-test loop.
Where Cycle-Time Gains Matter Most
- Early fit checks for patient-contact geometry
- Functional testing of clips, snap features, and instrument handling zones
- Design verification builds before tooling commitment
- Procedure simulation using patient-specific anatomical models
| Development Stage | Traditional Workflow | Additive-Enabled Workflow |
|---|---|---|
| Concept validation | Outsourced machining or hand-made mockups | Printed prototypes in days for immediate review |
| Design iteration | Long change cycles tied to tooling or setup | Digital revision-to-part loop with low change overhead |
| Pre-production verification | Limited builds due to cost and schedule pressure | Broader variant testing before release |
Surgical Planning and Training Models
Patient-specific anatomical models are now standard in many complex surgical planning workflows. They allow teams to rehearse approach, evaluate instrument access, and improve confidence before entering the operating room, particularly in cases with unusual anatomy.

Physical anatomical and mechanism models improve pre-procedure planning quality.
What Is Advancing Beyond Today's Baseline
Several emerging areas are expanding additive's role in medical technology. While readiness levels vary, they are increasingly relevant to long-horizon R&D and platform planning.
Bioprinting and Tissue Engineering
Bioprinting research is progressing toward structured tissue models for drug testing and disease modeling. Near-term impact is strongest in research environments, where printed biological constructs can improve model realism compared with conventional test systems.
Antimicrobial and Functional Surface Design
Material and surface engineering advances are enabling printed parts with active functionality such as antimicrobial behavior. If validated consistently, these approaches can reduce infection risk in specific device categories where surface hygiene is a primary concern.
Embedded Sensing and Smart Devices
Additive workflows are also being explored for integrating sensing features into device structures. The long-term value is continuous physiological monitoring without adding complex multi-part assemblies that increase manufacturing and validation burden.

Emerging medical additive programs are expanding from geometry to biological and sensing functionality.
How Teams Deploy Additive Responsibly
Successful adoption in medical contexts is rarely a printer-first decision. It is a workflow decision that links design controls, material selection, validation plans, and production quality systems.
Engineering Priorities
- Define clinical and mechanical requirements before process selection
- Match process capability to tolerance-critical features
- Design post-processing into the baseline workflow
Quality Priorities
- Establish inspection plans for patient-contact and fit-critical zones
- Document material controls and lot traceability
- Align verification evidence with regulatory submission needs
Conclusion
3D printing has already changed the economics and capability profile of medical device development. The strongest value appears where personalization, geometry complexity, and iteration speed intersect. For teams operating under clinical and regulatory constraints, additive manufacturing is most effective when treated as part of an integrated engineering system rather than a stand-alone fabrication option.
Evaluating a Medical Device Program for Additive Manufacturing?
Forge Labs supports engineering teams with material selection, process fit assessment, and production-ready workflows for regulated medical applications.
Speak With an Engineer